In a Heartbeat
What would it take to get back a heartbeat?
We hear about cardiovascular diseases (CVDs) all the time, with an estimated 31% of deaths globally1. It’s not uncommon to hear stories about a close friend who was young and had a heart attack. Perhaps your mom or dad had a stroke or even a neighbor who is having heart rhythm issues.
Every time I think about CVDs, the first thing that comes to my mind is my uncle. He was a healthy man in his 60s, very athletic, he used to swim and participate in marathons all the time. He never smoked, he always had a healthy diet and was active, and bam! One day, he had a stroke. To make the long story short, he ended up having to have a pacemaker implanted. He had a complication due to a pacemaker lead that was dispositioning, which caused a subsequent ischemic stroke.
Don’t take me wrong, pacemakers can be a good solution. However, complications like broken leads for instance can cause unnecessary “shocks” to the user. Complications that may occur during surgery include allergic reactions, infections, vessel damage, and heart tissue punctures.2 The weakest link in the pacemaker system, most often leading to complications, is the lead. This poses a question: Can we use an alternative source of energy to pace the heart? Can we use ultrasound as an alternative source of energy? Can we use ultrasound to treat CVDs?
If you would have a choice of another way to “control” the heart, would you do it in a heartbeat?
Cardiac Pacing
Looking back, if there was another way to pace my uncle’s heart and bring him back to a normal life, of course I would do it. Pacemakers work only when needed. If your heartbeat is too slow (bradycardia), the pacemaker sends electrical signals to your heart to correct the beat. The same principle is used if your heart is too fast (tachycardia). The pacemaker has a battery, and it works with electric energy. My goal in the last 10 years or so has been to use ultrasound as an alternative source of energy to pace the heart and to treat cardiovascular diseases.
The first step was to investigate if the ultrasound pulses can increase or decrease the heart rate (HR). Some of my preliminary studies3-5 showed that ultrasound applied to the heart of rats can cause a negative chronotropic effect (or decrease in the heart rate; Figure 1). The ultrasound protocol uses a sequence of different parameters, such as pulse duration (PD), pulse repetition frequency (PRF), etc. The PD is the distance each pulse travels and the pulse repetition frequency is the rate at which the transducer emits the pulses. The pulses must be spaced. This allows enough time between pulses, so the beam has enough time to reach the target and return to the transducer before the next pulse is generated. With a specific sequence, I was able to decrease the heart rate. Voila!



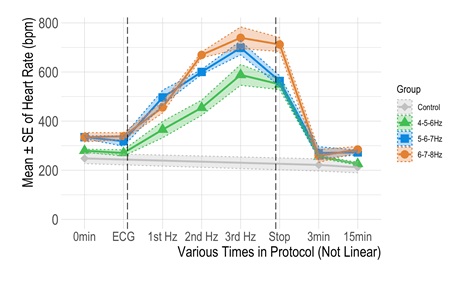
But what if I can increase the heart rate using ultrasound pulses? In a recent investigation, I was also able to increase the heart rate with a specific sequence of ultrasound pulses (Figure 2).
Is the Future Wireless?
We are all experiencing changes in gadgets in our daily life, from wireless vacuums to earplugs, chargers, etc. But can we pace the heart without leads? The question remains such as my uncle’s story. He’s still an active guy, but of course he needs to avoid problems like electromagnetic interference (cell phones, metal detectors, etc). Can we have a therapy that uses ultrasound to pace the heart, that is non-invasive, wireless, safe, and feasible? I hope so. Maybe one day I can change this scenario in a heartbeat!
References
1. World Health Organization. Fact Sheets: Cardiovascular Disease. Accessed: July 13, 2020. [Online]. Available: https://www.who.int/en/news-room/fact-sheets/detail/cardiovascular-diseases-(cvds)
2. Pakarinen S, Oikarinen L, and Toivonen L. Short-term implantation related complications of cardiac rhythm management device therapy: A retrospective single-centre 1-year survey. Europace 2010; 12:103–108. doi: 10.1093/europace/eup361.
3. Coiado OC and O’Brien WD. The negative chronotropic effect in rat heart stimulated by ultrasonic pulses: Role of sex and age. J Ultrasound Med 2017; 36:799–808.
4. Coiado OC and O’Brien WD. The role of the duty factor in ultrasound-mediated cardiac stimulation. J Acoust Soc Am 2014; 136:EL231–EL235.
5. Coiado OC, Buiochi E, and O’Brien W. Ultrasound-induced heart rate decrease: Role of the vagus nerve. IEEE Trans Ultrason Ferroelectr Freq Control 2015; 62:329–336.
6. Coiado OC, Yerrabelli RS, Christensen AP, Wozniak M, Lucas A, O’Brien WD Jr. Positive chronotropic effect caused by transthoracic ultrasound in heart of rats. JASA Express Lett 2021; 1:08200.
Dr. Coiado, PhD, is a Teaching Associate Professor in both the Carle Illinois College of Medicine and the Bioengineering Department at the University of Illinois at Urbana-Champaign. At Carle Illinois College of Medicine, she acts as the Director of Student Research and Discovery Learning. Her research interests focus on cardiovascular studies, acoustics, bioinstrumentation, and education.
Interested in reading more about vascular ultrasound? Check out these resources:
- JUM article by Coiado et al: The Negative Chronotropic Effect in Rat Heart Stimulated by Ultrasonic Pulses: Role of Sex and Age
- JUM article by Coiado et al: Therapeutic Ultrasound in Cardiovascular Medicine
- From the Scan: Ultrasound in Central Vein Assessment – The Importance of Knowing
- From the Scan: The Expanding Scope and Diagnostic Capabilities of Vascular Ultrasound
- From the Scan: How to Obtain Focused Cardiac Ultrasound Images













 2.
2.  3.
3.  4.
4.  5.
5. 









